- To generate electric current, two particles must separate in spite of being bound together closely.
- These two particles, electron and electron-hole, together form an exciton.
- The higher mobile excitons can achieve more efficient charge separation.
Physicists at Purdue University have discovered a method for creating a charge in organic solar cells – a long-lived physics puzzle. Their method includes using an electron-hole interface, in which electrons and holes can be close together or far apart.
Organic solar cells are usually made of soft molecules, most of them are polymer solar cells, while inorganic solar cells, mostly based on silicon, are developed with more rigid material. There is no doubt that silicon cells are currently dominating the industry, but they are stiff and expensive.
The molecules used in organic solar cells, on the other hand, are flexible and solution-processable at high throughput. And since they are cheap, the production cost of fabricating a large volume is significantly low. The only negative point is, and it’s a major one, generating electric current in organic cells is an extremely difficult task.
Electron-Hole Interface
To generate electric current, two particles must separate in spite of being bound together closely. These two particles, electron (negative charge) and electron-hole (positive charge), together form an exciton. In order to separate these particles, you need an interface that draws the electron via an electron acceptor, leaving behind a hole.
The problem is not solved here – even if you get the interface in the right place, the electron and hole are still attracted to each other.
The research team found out that these electron-hole interfaces are not one single static state. They could be close to each other or far apart. The farther they’re from each other, the more likely they’re to separate.
Also, when electron and holes are far apart, they are very mobile and can move fast. This type of fast motion between the negative and positive charge drives separation at these interfaces.
Reference: Sciencemag | doi: 10.1126/sciadv.aao3104 | Purdue University
UltraFast Microscopy
Since organic solar cells are quite messy, they’re hard to study. It’s even harder to implement optical spectroscopy at that size-scale. Their states don’t exist for long duration, so you need a very short time resolution.
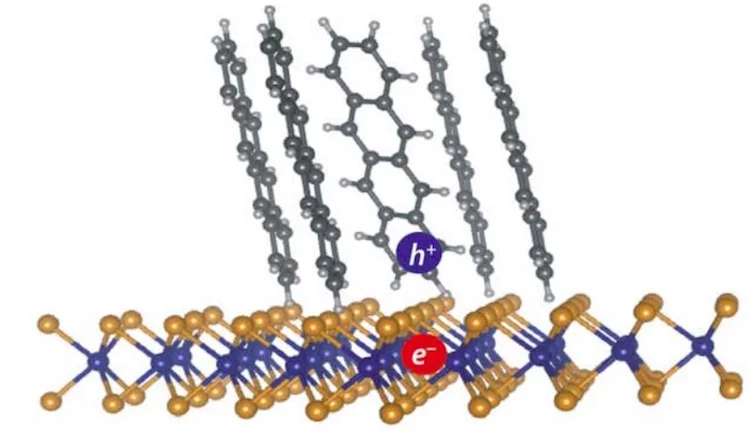 Exciton formed at the interface between an organic (tetracene molecules) and inorganic semiconductor (single-layer WS2)
Exciton formed at the interface between an organic (tetracene molecules) and inorganic semiconductor (single-layer WS2)
Researchers have built a tool named ultrafast microscopy to observe the processes (by merging time and spatial resolution) that lasts for very short duration. However, the spatial resolution still wasn’t good enough. So they built a bigger, 2D interface to create order in the molecules’ chaotic arrangement. Therefore, the problem has two-fold solution – the interface and ultrafast microscopy.
More specifically, the charge-transfer excitons at heterointerfaces play an important role in conversion of light to electricity using nanostructured and organic material. They studied the formation and transportation of these exciton in 2D tetracene heterostructures.
Electron and hole transfer take place for a few picoseconds (10−12 seconds), and inter-layered excitons emit with a binding energy of approximately 0.3 eV. Excitons’ transport is calculated by transient absorption microscopy, unveiling coexistence of localized and delocalized states.
Trapping-and-detrapping dynamics between the localized and delocalized states leads to stretched exponential photoluminescence decay with an average lifetime of nearly 2 nanoseconds. The delocalized excitons are exceptionally mobile with about 1 cm2 s-1 diffusion constant. The higher mobile excitons can achieve more efficient charge separation.
Read: A New Type Of Solar Cells That Can Be Washed and Stretched
Having a knowledge of how excitons separate could help physicists develop new interfaces for organic solar cells. This means materials to develop solar cells with that have not been utilized yet.


